Among patients transported during the 7-year study period, we identified 2,157 trauma patients with prehospital lactate values (Figure 1) obtained according to the blood administration protocol (Supplementary Table 1 and Appendix 2).

Flow diagram showing cohort selection, with STROBE guidelines indicated in grey boxes. Injured subjects were admitted to a trauma or burn unit or experienced the following mechanisms of injury: assault, animal bite, burn, electric shock (other than lightning), gunshot wound, stab/cut wound, machinery accident, pedestrian, bicycle, motor vehicle, 4×4, motorcycle, water transport, or aircraft accident, crash, or collision.
Of the cohort, 1,480 (68.6%) patients were male, the mean age was 47 years (IQR = 28 – 62), and 207 (9.60%) patients had the primary outcome of requiring a blood transfusion within 4 hours of emergency department admission (Table 1).
The median prehospital lactate concentration was 4.85 mmol/L (IQR = 2.30–5.80) in subjects who received blood products and 2.48 mmol/L (IQR = 1.30–2.98) in subjects who did not require hospital blood products within 4 hours of admission. Of subjects who received hospital blood products, 19 (10%) died within 24 hours of admission. Of subjects who did not require hospital blood products, only 1% died within 24 hours of admission (yeah = 15). Consistently, a higher proportion of subjects who received hospital blood products required life-saving interventions (LSIs) in other hospitals (Table 1).
Information on prehospital blood and crystalloids provided by prehospital care services and before arrival was excluded from the decision process due to significant collinearity (i.e. relationships between model predictors) associated with blood administration within the hospital. We provided the FFT algorithm with 13 variables that could be selected based on clinical value and availability to prehospital clinicians. [12]Among these was the AIS score, which was offered as a surrogate for trauma status that prehospital clinicians could see and which we have previously found to be associated with blood transfusion in the hospital. We acknowledge that AIS values are not available in the prehospital environment, but we use it here as a surrogate for clinically recognizable anatomical trauma patterns used by EMS clinicians in the field for trauma triage purposes. Five of the 13 variables were not selected by the algorithm because they were not associated with the need for blood transfusion: 1) severely elevated heart rate > 120 bpm, 2) abdominal AIS > 2, 3) spinal AIS > 2, 4) type of trauma (blunt or penetrating), and 5) shock index (SI) range (difference between highest and lowest SI).
The algorithm generated four variables that were highly associated with hospital transfusion within 4 hours of admission (Figure 2). The variables selected by the algorithm were evaluated in the following order: 1) minimum SBP (continuous), 2) pre-admission venous lactate (continuous), 3) minimum SI (continuous), and 4) AIS chest > 2 (categorical). Predictors not selected by the algorithm were: Fast Fourier Transform The algorithm requirements were 1) age, 2) mission type (on-site or interfacility transfer), 3) AIS head > 2, and 4) AIS lower extremities > 2. This FFT had a sensitivity of 0.81 and a specificity of 0.71 based on a data-driven variable sequence and thresholds.

Pilot FFT selected by the algorithm. The top panel shows the number of observations and the outcome (4 hours of hospital blood administration). SBP_min – minimum SBP (mmHg), nlacven – pre-admission lactate concentration (mmol/L), si_min – minimum SI (bpm/mmHg), ais_ab – abdominal AIS (0/1, equals 1 if AIS > 2). A “hit” (green triangle) indicates a correct blood administration and a “miss” (red triangle) indicates an incorrect rejection. Sensitivity (triangle) = hit / (hit + miss). A correct rejection (green circle) indicates a correct decision. do not haveDonate blood,false alarms (red circles) mean false positives, i.e., incorrect decisions to donate blood.,Specificity (circles) = number of correct rejections/(number of correct rejections + number of false alarms).,Pilot FFTs were obtained using the training and test,datasets (test dataset). no= 1,121), the tree “fan” of possible trees was selected that offered the best balance between sensitivity and specificity. The default sensitivity weight of 0.5 resulted in a “zigzag” shape with alternating decisions. The ROC panel shows the resulting FFT and a comparison of parameters with other common model building approaches: CART (C, red), logistic regression (LR, blue), random forest (RF, purple) and support vector machine (SVM, yellow).
We applied the FFT definition from the pilot experiment with rounded thresholds to the entire study population, with similar performance (Supplementary Figure 1A, sensitivity = 0.84, specificity = 0.70). We then maximized the sensitivity parameter, with the goal of administering hospital blood to the maximum number of eligible patients while minimizing erroneous administration. Setting the weighting parameter to any value in the range of 0.7–1 resulted in a “positive rake” FFT that made a positive blood decision after every node (Supplementary Figure 1B, sensitivity = 0.93, specificity = 0.39). We also see from Supplementary Figure 1B that the positive predictive value (PPV) of the model is 14.0% (192/1,373), whereas the negative predictive value is 98.1% (769/784).
The resulting FFT outperformed other model-building approaches (e.g., CART and logistic regression (LR)) to create a decision-support model for early hospital blood administration with higher sensitivity and specificity (Supplementary Fig. 1B).Finally, we modified and simplified the tree definition using current field triage guidelines and traditional thresholds used in the literature to account for its potential use in the prehospital setting. [13].
The FFT algorithm found variable thresholds that differed from the traditional ones (Figure 2). We investigated thresholds that were already commonly used (i.e., SBP threshold 90 mmHg and EMS lactate 4 mmol/L) or that were easy for EMS personnel to calculate (SI > 1 = HR > BP). [14]When applying traditional thresholds (Supplementary Figure 1C) instead of data-driven thresholds (Supplementary Figure 1B) to our dataset, the sensitivity is significantly reduced but the specificity parameters are increased. We tested (a) how changing the FFT definition with traditional thresholds affects the sensitivity and specificity parameters (Supplementary Figure 1C, Table 2, first blue row) and (b) whether specificity and sensitivity can be balanced by using a combination of traditional and newly discovered thresholds (Table 2, yellow row). The tree was created in the same way as in Supplementary Figure 1B (Table 2, first row), where only the thresholds are different (FFT thresholds and parameters in Supplementary Figure 1B are highlighted in orange in Table 2). Table 2 shows how varying thresholds for SBP, lactate, and shock index change the sensitivity, specificity, and overall performance based on Youden's J index. As expected, higher SBP, lower lactate, and lower SI thresholds increase sensitivity but decrease specificity.
We recognized that pre-admission lactate may not be widely available and performed sensitivity analyses by removing the lactate term from the models and comparing FFT-derived thresholds with traditional thresholds for SBP and SI (Table 2, rows 2.1–2.4). Models including the lactate term often had higher sensitivity (compare rows 2.1 vs. 1.1/3, 2.2 vs. 1.2/4, 2.3 vs. 1.5/7, and 2.4 vs. 1.6/8) but lower specificity and Youden's J index.
We also assessed the current practice of prehospital transfusion by critical care services and the need for in-hospital early transfusion. Table 3 shows a cross-tabulation of actual prehospital blood administration by early prehospital transfusion. Of the 207 patients who required a prehospital transfusion, 79 (38.2%) had also received a blood transfusion before arriving at the hospital (Table 3, upper left quadrant). The majority (73) of these 79 patients had a systolic blood pressure <90 mmHg and received prehospital blood according to the transfusion protocol of the prehospital care service. Of the 60 patients who received a prehospital transfusion but did not require hospital blood (Table 3, upper right quadrant), 33 (55.0%) patients had a systolic blood pressure <90 mmHg. Patients who received a transfusion with a systolic blood pressure >90 mmHg received a transfusion on medical advice or off-protocol.
In our Bayesian analysis, the model with the most predictive power showed statistically significant associations of tachycardia (OR = 1.74, 95% CI 1.12–2.55), elevated prehospital lactate (OR = 2.31, 95% CI 1.55–3.37), and hypotension (OR = 11.59, 95% CI 7.70–16.98) with early in-hospital transfusion. In Bayesian subgroup analysis of patients with SBP > 90 mmHg (N = 1,901, 87.6%), the most predictive model included minimal shock index (OR = 25.6, 95% CI 2.54-113.2), elevated lactate (OR = 2.17, 95% CI 1.11-3.77), and tachycardia (OR = 1.59, 95% CI 0.72-2.94). Based on 95% confidence intervals, lactate and minimal shock index were significantly associated with higher posterior odds of early in-hospital transfusion in the hypotensive cohort.
By integrating and operationalizing the results of our approach for use in the field, we developed an algorithm for prehospital transfusion that incorporates prehospital SBP, prehospital lactate, shock index, and severe abdominal injury (Figure 3), with different thresholds that can be adjusted depending on system resources and time considerations.
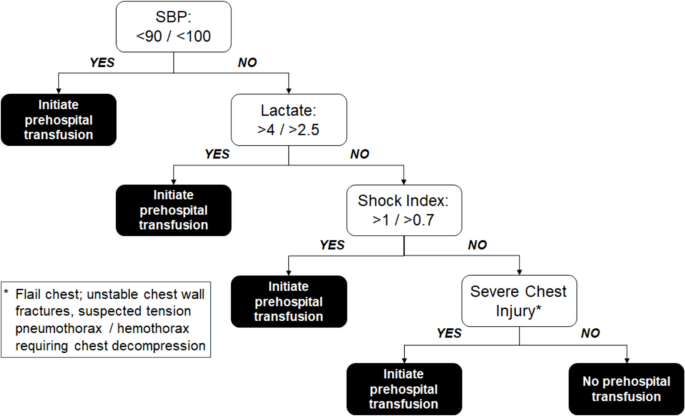
Summary of the derived decision rules and how they aid in prehospital transfusion. The rules were derived based on the need for in-hospital transfusion over a 4-hour period.
We also applied the FFT definition in Supplementary Figure 1B but excluded nodes with severe chest injury (Supplementary Figure 2). The resulting sensitivity and specificity parameters were slightly lower than those of the four-factor model (Supplementary Figure 1B, Supplementary Figure 2).



