Just a few years ago, we predicted how a series of amino acids incorporated into functional proteins was one of the most frightening puzzles in biology. That was until AI opened up and cracked the problem wide, winning global acclaim and last year's Nobel Prize in Chemistry.
That breakthrough relied on deep learning, a form of AI that learned from large data sets and predicted protein shapes and more. Now, researchers are based on their momentum with generative AI. From unknown protein sequences to novel treatments for a wide range of diseases, a powerful new class of AI models that allow you to imagine completely new possibilities rather than predicting structure.
“Most protein design work begins with a fully experimentally determined 3D map of the target,” says Timothy Jenkins of the Institute of Technology, Denmark. 'But for many of the most important things [therapeutic] Targets… these maps simply do not exist. This is a major obstacle for personalized medicine. ”
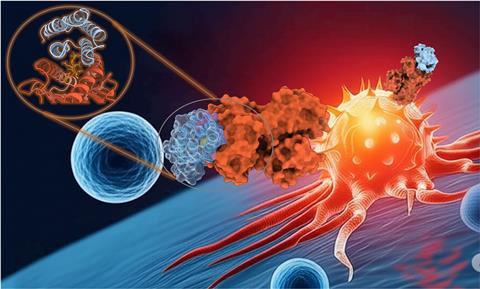
Jenkins and his collaborators recently published a study using a generator AI model called RFDiffusion to design molecules that help the immune system recognize and attack cancer.1 “Simply put, we created a new, ultra-fast pipeline to create precision cancer immunotherapy,” he says.
Targeted Therapy Game Changer
This process works like a fast assembly line where various AI tools handle different steps. “First, we'll show rfdiffusion a 3D image of the cancer flag you want to target,” explains Jenkins. This 3D image can be obtained from experimental data or from AI-based structural predictions. “AI can then “dream” thousands of completely new small protein forms.
The second AI model takes their shapes and grasps the exact amino acid sequence needed to construct them. “The process is very fast,” says Jenkins. “It can take hours or days to generate thousands of potential designs on a computer, but it can take months or years to find a candidate using traditional lab methods.”
Molecular dynamics simulations are then used as “virtual crash tests” to see how well the designed proteins stick to cancer targets and help teams narrow down candidates before moving on to lab experiments.
“The fact that we were able to create successful binders based purely on computational predictions is a game changer,” Jenkins says. “Our approach is not limited to the number of well-characterized targets and shows that it can be expanded to design treatments for truly individualized cancer targets where structural information is not available.”
Dreaming of proteins to kill deadly bacteria
Beyond personalized cancer therapy, a group led by RHYS Grinter at the University of Melbourne and Gavin Nott at Monash University use AI produced to design proteins that kill antibiotic-resistant bacteria.2
“AI-based protein design allowed us to dream of being the small protein that connects chua,” says Grinter. Chua is an outer membrane transporter protein used in pathogenic bacteria E. coli and Sozumi Extract heme, a rich source of iron from the host.
Using Alphafold2, the team predicted the 3D structure of the chua from its amino acid sequence in minutes. A strategy was then developed to evaluate the accuracy of the model and block Chua's functionality. It was then used to design proteins that could interfere with the target using the generator AI tools, Rfdiffusion, and ProteinMpnn.
“The whole process took several weeks and there was a 10-50% success rate in functional design,” says Knott. “This approach effectively shaves months to years from standard experimental structural biological approaches, and rapidly accelerates the development of new biology.”
“It's amazing and highlights the power of AI and the potential for transformation in protein design,” adds Glitter.
Grinter, Knott, Jenkins and their colleagues prefer AI platforms that are freely accessible to the wider scientific community. Making all of these tools available has helped reduce barriers for researchers around the world. As a result, a wide range of experts can work together to innovate and promote progress that imposes social challenges such as antibiotic resistance and personalized cancer therapy.
“But it's not as simple as point and click,” says Knott. “At this point, this technology still requires a deeper understanding of protein structure and functional relationships.”
And despite the impressive capabilities of generative AI, there are inherent limitations rooted in the data it learns, so caution is still needed. “The Generator AI is very powerful, but it's not magic,” says Jenkins. “Models are trained on thousands of protein structures we already know. When asked to design something fundamentally different from what we “see” before, we can “hapticize” designs that look good on computers but are not physically stable or functional in the real world. ”
“Because biology exists in the real world, AI achieves true value when applied to biological systems in conjunction with experimental technology,” concludes Grinter. “Combining experiments and AI gives us the power of transformation for scientific discovery and technological development. We believe that in the future AI tools will form the central element of almost every aspect of biological research.




