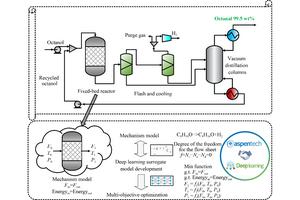
The selective oxidation of alcohols to aldehydes is an important reaction in organic synthesis and chemical production. However, this reaction exhibits a complex reaction network that is influenced by temperature, time, and pressure conditions. A study published in Frontiers of Chemical Science and Engineering presents a hybrid modeling strategy to address optimization challenges through artificial intelligence-assisted process optimization.
The research team focused on the oxidation process of isooctanol. In this process, iso-octanol mixed with unreacted iso-octanol flows into a fixed bed reactor, producing iso-octanal and hydrogen byproducts. The gas-liquid mixture is cooled and separated in two flash tanks. The liquid product enters the vacuum distillation column, yielding 99.5% pure iso-octanol at the top, and unreacted iso-octanol is recycled to the reactor.
To overcome the lack of experimental data due to long operating cycles and hydrogen safety concerns, the researchers used Aspen Plus to build an accurate mechanistic model based on existing research data. The PENG-ROB equation of state was used for physical property calculations, and a plug flow reactor was selected as the reactor. Once the mechanistic model converges, the calculated data satisfies the material and energy balance. This mechanistic model generated 1,845 sets of reliable training data.
Four machine learning models were investigated as surrogate models: deep neural networks, deep belief networks, extreme gradient boosting, and random forests. These alternative models were integrated with four multi-objective optimization algorithms, including NSGA-II, NSGA-III, MOEA/D, and RVEA, to identify optimal operating conditions that balance production costs and carbon emissions.
The results demonstrated significant efficiency gains. The surrogate model achieved computational speeds over 400 times faster than traditional direct simulation methods. Among the 16 optimization model combinations tested, the combination of XGBoost and RVEA showed the fastest calculation speed, achieving a short solution time of 14.3 seconds.
The optimization results revealed significant environmental benefits. After optimization, the primary energy demand was reduced by 10% to 5.09 gigajoules per hour. A significant 12% reduction in greenhouse gas emissions was achieved, reaching 0.29 tons of carbon dioxide equivalent per hour.
Further analysis showed that the main energy consuming sub-processes after optimization were the dehydrogenation reactor and distillation separation unit, accounting for 54 and 46 percent, respectively. Carbon dioxide equivalents account for the majority of greenhouse gas emissions, accounting for 64 percent and 36 percent, respectively.
Correlation analysis using the SHAP interpretability method revealed that reaction temperature has a greater impact on both total production value and greenhouse gas emissions than other variables such as residence time, reaction pressure, number of distillation trays, and feedstock location. In particular, reaction temperature showed a strong negative correlation with production cost, suggesting that increasing temperature increases the evaporation rate of the feedstock to the distillation column and reduces separation costs.
This study provides impetus for the engineering application of the octanol dehydrogenation reaction to octanol production and serves as a reference for the industrial application of the alcohol dehydrogenation reaction to high-value aldehydes.
Disclaimer: AAAS and EurekAlert! We are not responsible for the accuracy of news releases posted on EurekAlert! Use of Information by Contributing Institutions or via the EurekAlert System.